NEJM: Multistate Infant Botulism Outbreak Associated with Powdered Infant Formula

Authors: Connie H. Chung, M.P.H., Jason R. Barash, Jacquelin L. Castonguay, M.P.H., Claire Penzel-McNamara, M.P.H., Katie Lee, Ph.D., Jennifer S. Read, M.D., M.S., M.P.H., D.T.M.&H., Kerry A. Padgett, Ph.D., and Jessica M. Khouri, M.D.
Abstract
This report details the identification of an outbreak of infant botulism linked to powdered infant formula in the United States. In October 2025, the Infant Botulism Treatment and Prevention Program, California Department of Public Health (IBTPP-CDPH) noted common powdered infant formula exposure among three neonates with suspected infant botulism. Subsequent laboratory analyses at CDPH identified Clostridium botulinum type A from an open container of ByHeart powdered infant formula associated with one infant with infant botulism. As of December 10, 2025, 51 suspected or confirmed infant botulism cases with exposure to ByHeart powdered infant formula had been identified across 19 states. All ByHeart powdered infant formula has been recalled nationwide.
Introduction
Infant botulism is a rare, life-threatening disease that occurs when swallowed spores of Clostridium botulinum germinate, colonize, and produce botulinum neurotoxin (BoNT) in an infant’s large intestine, resulting in descending flaccid paralysis.1 Treatment is based on clinical diagnosis2 and should not be delayed for laboratory testing. Human Botulism Immune Globulin Intravenous (BIG-IV)3 is the only Food and Drug Administration (FDA)–approved treatment for infant botulism caused by BoNT types A and B (responsible for 99% of infant botulism in the United States). BIG-IV is sponsored, produced, and distributed by the Infant Botulism Treatment and Prevention Program (IBTPP) at the California Department of Public Health (CDPH).4 Deployment of BIG-IV serves as a surveillance mechanism to identify cases nationally. Cases of infant botulism typically occur sporadically; it is not usually a disease that causes outbreaks. Soil-dwelling clostridial species have been identified in samples of powdered infant formula,5and a case of infant botulism was associated with powdered infant formula in the United Kingdom.6 IBTPP routinely collects information on consumed products for all treated patients.
ByHeart launched sales of powdered infant formula in March 2022. The first report of ByHeart powdered infant formula consumption by an infant in whom infant botulism developed was in 2023; fewer than five cases reported exposure during 2023 and 2024 combined. Although additional cases of infant botulism caused by both BoNT type A and B in 2025 reported exposure to ByHeart powdered infant formula, toxin type distribution aligned with expected geographic norms, and a point source outbreak was not suspected.
Investigation and Outcomes
In late October 2025, the IBTPP deployed BIG-IV to three hospitalized neonates who had consumed ByHeart powdered infant formula prior to illness onset. One was a California neonate whose stool specimen tested positive for BoNT type A at CDPH Microbial Diseases Laboratory (MDL), Botulism Reference Unit (BRU), confirming the diagnosis of infant botulism. The family requested testing of the opened containers of ByHeart powdered infant formula from which the infant had been fed prior to onset. Two opened formula containers were received at BRU and tested using methods developed in-house.5 The enrichment culture of one container tested positive for BoNT type A. C. botulinum type A was isolated from subcultures of the powdered infant formula enrichment culture containing BoNT type A. Whole-genome sequencing of isolates from the patient’s stool and associated formula were found to be C. botulinum subtype A1.
In parallel, the IBTPP received laboratory reports confirming type A infant botulism for two patients treated in Pennsylvania and Rhode Island. Whole-genome sequencing of the isolate from the Rhode Island patient’s stool revealed C. botulinum subtype A1. Detection of type A is rare east of the Mississippi River. This was the first type A case identified in Rhode Island, and 98% of cases in Pennsylvania are type B. Case investigation by IBTPP noted ByHeart powdered infant formula exposure in both patients.
ByHeart’s small projected market share, the number of suspected infant botulism cases with common exposure to the product, including among neonates, and the atypical geographic distribution of toxin types led IBTPP to consider whether there was a potential association between ByHeart powdered infant formula and infant botulism. IBTPP contacted the Centers for Disease Control and Prevention (CDC) and the FDA on November 5, 2025, about the common formula association. On November 7, 2025, ByHeart powdered infant formula was named as the common exposure, and the decision was made by CDC and FDA to mobilize operational response for a multistate outbreak, including outreach to the firm. At this point, there were 13 suspected cases of infant botulism with reported exposure to ByHeart powdered infant formula.
On November 8, 2025, ByHeart recalled two lots of powdered infant formula associated with recent cases. On November 11, 2025, ByHeart voluntarily recalled all infant formula products following the identification of additional suspected patients with infant botulism, expanded lot code associations, and reporting of the preliminary positive result for C. botulinum type A in the powdered infant formula sample tested by CDPH MDL. ByHeart partnered with IEH Laboratories to test unopened product. On December 23, 2025, the firm reported positive test results for Clostridium botulinum in six of 36 samples of finished product from two lots.
CDC, FDA, and IBTPP codeveloped the outbreak case definitions (Table 1). On December 10, 2025, CDC expanded the timeframe of the case definitions to include all confirmed or suspected cases with ByHeart powdered infant formula exposure going back to March 23, 2022 (when the formula became available).
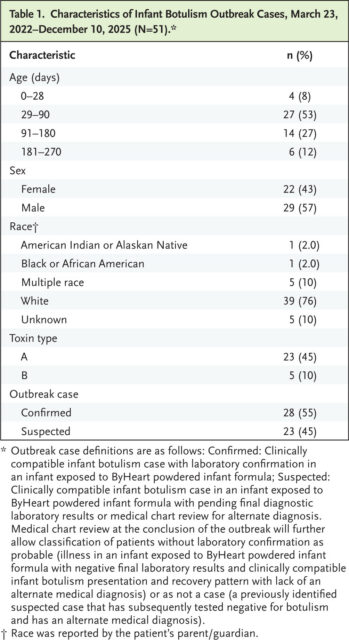
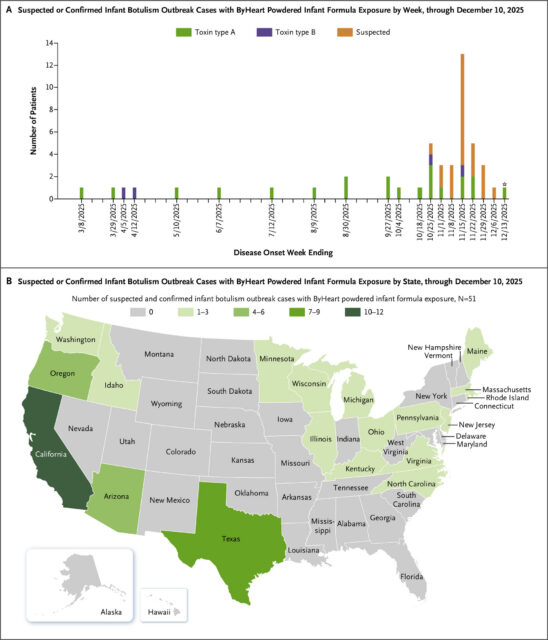
Suspected or Confirmed Infant Botulism Outbreak Cases with ByHeart Powered Infant Formula Exposure through December 10, 2025 (N=51).
Characteristics of Infant Botulism Outbreak Cases, March 23, 2022–December 10, 2025 (N=51).
As of December 10, 2025, 51 cases of suspected or confirmed infant botulism and exposure to ByHeart powdered infant formula had been reported across 19 states (Table 1 and Fig. 1). All patients required hospitalization. No outbreak-associated deaths have been reported. IBTPP, CDC, and state and local health departments are continuing their investigations, facilitating BIG-IV treatment, diagnostic stool testing, and obtaining and testing containers of powdered infant formula associated with infant botulism cases. Testing and whole-genome sequencing analysis of unopened powdered infant formula and whole milk powder by FDA showed genetic matches between whole milk powder, finished powdered infant formula product, and an infant botulism clinical isolate.7 These findings indicate that consumption of ByHeart powdered infant formula resulted in this multistate infant botulism outbreak. Testing of additional powdered infant formula products and ingredients and whole-genome sequencing analyses continue. FDA is working with state partners to facilitate removal of products from all retail locations. These events constitute a multistate infant botulism outbreak associated with powdered infant formula in the United States.5,6
REFERENCES
- Committee on Infectious Diseases, American Academy of Pediatrics. Botulism and infant botulism (Clostridium botulinum). In: Kimberlin DW, Banerjee R, Barnett ED, Lynfield R, Sawyer MH, eds. Red book: 2024–2027 report of the committee on infectious diseases. Itasca, IL: American Academy of Pediatrics, 2024:308-312.
- Rao AK, Sobel J, Chatham-Stephens K, Lúquez C. Clinical guidelines for the diagnosis and treatment of botulism, 2021. MMWR Recomm Rep 2021;70:1-30.
- Arnon SS, Schechter R, Maslanka SE, Jewell NP, Hatheway CL. Human botulism immune globulin for the treatment of infant botulism. N Engl J Med 2006;354:462-471.
- Payne JR, Khouri JM, Jewell NP, Arnon SS. Efficacy of human botulism immune globulin for the treatment of infant botulism: the first 12 years post-licensure. J Pediatr 2018;193:172-177.
- Barash JR, Hsia JK, Arnon SS. Presence of soil-dwelling clostridia in commercial powdered infant formulas. J Pediatr 2010;156:402-408.
Republished with permission from Bill Marler and Marler Clark. Copyright (c) Marler Clark LLP, PS. All rights reserved.
Source: https://www.marlerblog.com/case-news/nejm-multistate-infant-botulism-outbreak-associated-with-powdered-infant-formula/
Anyone can join.
Anyone can contribute.
Anyone can become informed about their world.
"United We Stand" Click Here To Create Your Personal Citizen Journalist Account Today, Be Sure To Invite Your Friends.
Before It’s News® is a community of individuals who report on what’s going on around them, from all around the world. Anyone can join. Anyone can contribute. Anyone can become informed about their world. "United We Stand" Click Here To Create Your Personal Citizen Journalist Account Today, Be Sure To Invite Your Friends.
LION'S MANE PRODUCT
Try Our Lion’s Mane WHOLE MIND Nootropic Blend 60 Capsules
Mushrooms are having a moment. One fabulous fungus in particular, lion’s mane, may help improve memory, depression and anxiety symptoms. They are also an excellent source of nutrients that show promise as a therapy for dementia, and other neurodegenerative diseases. If you’re living with anxiety or depression, you may be curious about all the therapy options out there — including the natural ones.Our Lion’s Mane WHOLE MIND Nootropic Blend has been formulated to utilize the potency of Lion’s mane but also include the benefits of four other Highly Beneficial Mushrooms. Synergistically, they work together to Build your health through improving cognitive function and immunity regardless of your age. Our Nootropic not only improves your Cognitive Function and Activates your Immune System, but it benefits growth of Essential Gut Flora, further enhancing your Vitality.
Our Formula includes: Lion’s Mane Mushrooms which Increase Brain Power through nerve growth, lessen anxiety, reduce depression, and improve concentration. Its an excellent adaptogen, promotes sleep and improves immunity. Shiitake Mushrooms which Fight cancer cells and infectious disease, boost the immune system, promotes brain function, and serves as a source of B vitamins. Maitake Mushrooms which regulate blood sugar levels of diabetics, reduce hypertension and boosts the immune system. Reishi Mushrooms which Fight inflammation, liver disease, fatigue, tumor growth and cancer. They Improve skin disorders and soothes digestive problems, stomach ulcers and leaky gut syndrome. Chaga Mushrooms which have anti-aging effects, boost immune function, improve stamina and athletic performance, even act as a natural aphrodisiac, fighting diabetes and improving liver function. Try Our Lion’s Mane WHOLE MIND Nootropic Blend 60 Capsules Today. Be 100% Satisfied or Receive a Full Money Back Guarantee. Order Yours Today by Following This Link.






